|
Two scientists are credited with independently discovering bromine, Carl Jacob Lowig in 1825 and Antoine Balard in 1826. Polyvinyl chloride (PVC or vinyl) plastic pipes.Uses:Ĭhlorine compounds have a very large number of applications in industry in the production of organic compounds, plastics like polyvinyl chloride (PVC), for precursors used in the purification of metals, and as bleaches and disinfectants, such as in the sanitation of drinking water. The alternative name halogen was proposed, meaning salt-producer, but instead came to refer to the group to which chlorine belongs. The following year Sir Humphry Davy concluded that it is an element, which he named chlorine from the Greek chloros meaning pale green-yellow, which is the colour of chlorine gas. In 1809, Joseph Gay-Lussac and Louis-Jacques Thenard tried to release muriaticum from muriatic acid air and failed, raising the possibility that it was itself an element. Scheele failed to recognise it as an element and it was eventually believed to be an oxide of the hypothetical element muriaticum. Polytetrafluorethylene (PTFE or Teflon) tape, tube used in medical devices, and rod.Ĭhlorine compounds like salt have been used since ancient times, and chlorine gas was produced from the 1200s as a by-product of acid production, however its discovery as an element began in 1774 when Carl Wilhelm Scheele produced chlorine gas and called it dephlogisticated muriatic acid air.Fluorine is also used in uranium enrichment and in refrigerants (CFCs) though the latter have been phased out as they damage the ozone layer. Fluorine salts are used in tap water and toothpaste to prevent tooth decay. The fluorine-containing polymer PTFE (Teflon) is used in electrical insulation, medical devices and cookware. Uses:įluorine compounds are still used in the processing of metals. Although experiencing serious poisoning, Henri Moissan was the first to successfully isolate fluorine gas in 1886. Several scientists then attempted to isolate fluorine from hydrofluoric acid, but were either injured or killed by it, as it is a very toxic substance. 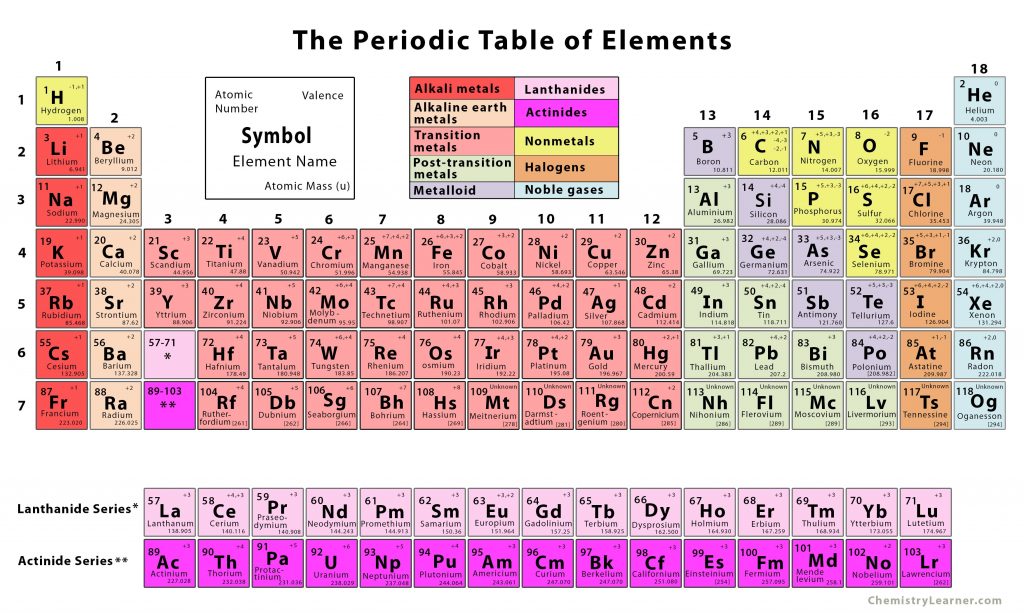
Andre Marie Ampere proposed in 1810 that this acid consisted of hydrogen and another element similar to chlorine, and Sir Humphry Davy named this new element fluorine, combining fluo- from the name of the acid and the -ine suffix used in naming halogens. In the 18th century hydrofluoric acid (then called fluorspar acid), made from fluorite, was used in glass etching. It was named fluores after the Latin word fluor, meaning flow. What elements are in Group 17 – Halides?Ĭlick on the following elements to learn more about them:įluorite (CaF 2) was used in the 16th century in the processing of metals as an additive to lower their melting points and make them easier to process. Therefore, there are various non-equivalent definitions of atomic radius.We take your privacy seriously. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. 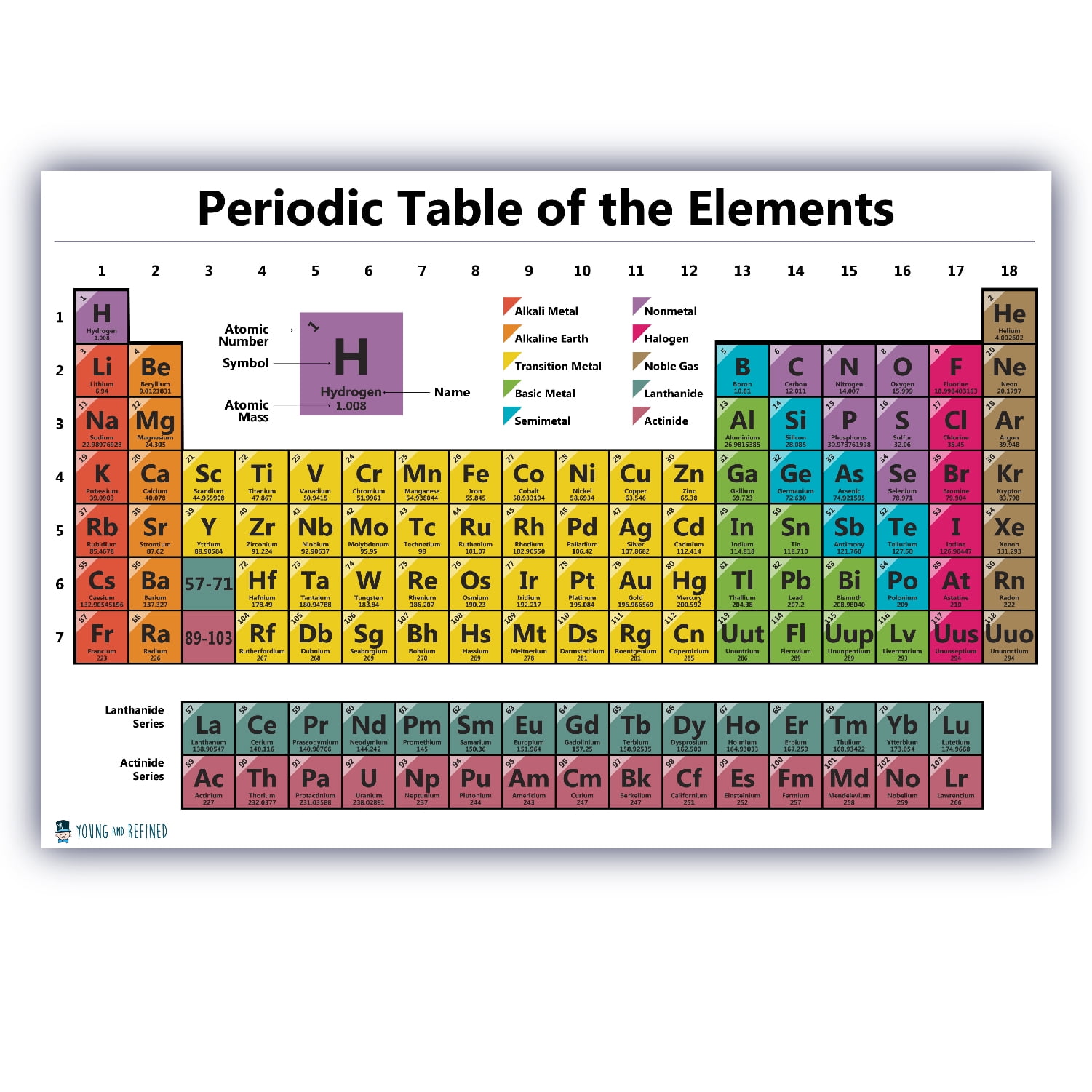
The atomic radius of Oxygen atom is 66pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Oxygen are 16 17 18. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. 
The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Oxygen is a chemical element with atomic number 8 which means there are 8 protons in its nucleus. Atomic Number – Protons, Electrons and Neutrons in Oxygen
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
